In the absence of flavorful ingredients, well-browned food has a richer, more varied flavor then poorly browned food.
What is browning?
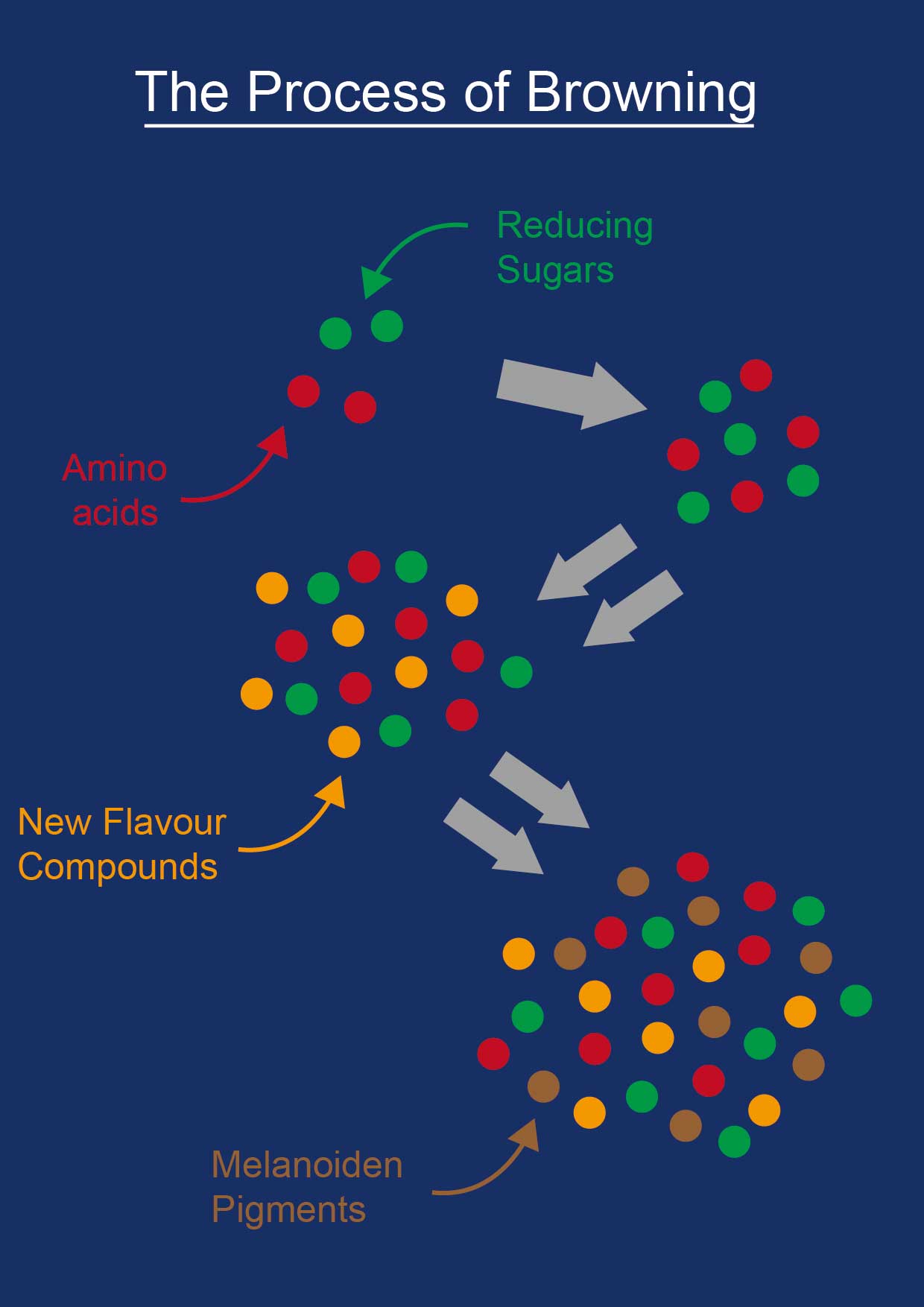
 Browning is a chemical reaction, also known as the Maillard Reaction after the French scientist, Louis Camille Maillard, who first described the process in the early 1900s. The chemistry behind this reaction is not completely understood even today but, put simply, in many foods heat causes the amino acids (the building blocks of proteins) to react with certain types of sugars to create new, distinct flavor compounds. These compounds in turn react with more amino acids to form even more compounds, rapidly increasing in quantity on both the surface of the cooking food and the pan itself. Finally, very large molecules called Melanoidin pigments are formed which create the deep brown hue on the crusts of meat and vegetables.
Browning is a chemical reaction, also known as the Maillard Reaction after the French scientist, Louis Camille Maillard, who first described the process in the early 1900s. The chemistry behind this reaction is not completely understood even today but, put simply, in many foods heat causes the amino acids (the building blocks of proteins) to react with certain types of sugars to create new, distinct flavor compounds. These compounds in turn react with more amino acids to form even more compounds, rapidly increasing in quantity on both the surface of the cooking food and the pan itself. Finally, very large molecules called Melanoidin pigments are formed which create the deep brown hue on the crusts of meat and vegetables.
Sauteing and searing are generally considered the best cooking methods for giving meat, such as steak, or vegetables a well-browned, flavourful crust. In order to achieve this, a proper pan temperature is crucial for success. If the pan is too cool the foods may cook too slowly and virtually steam in exuded juices, preventing both crust and flavor development. The Maillard Reaction occurs only when the surface temperature exceeds 300 degrees.
What is the best frying pan for a delicious, browned crust on food?
Although aluminum and tri-ply stainless steel pans conduct heat around a frying pan very evenly, they do not conduct it to the surface of the pan very quickly. Significant heat is required to jump start the chemical reaction that causes food to brown; and longer cooking time, as with an aluminum or stainless steel pan, won’t lead to more browning unless the food is cooked for a very long time. However, if food is cooked for too long there will be large loss of moisture, so that that although the food might be well-browned on the outside, it will be too dry and chewy on the inside.
Ultimately, a hotter pan equals more browning. Cast iron is excellent for this because it not only retains heat extremely well but also, once heated up, conducts heat to the surface of the food very quickly. However, for cooking usage cast iron does have some significant disadvantages: it is heavy, expensive, difficult to clean, and the temperature of the pan can be difficult to control.
In contrast, a Lotus Rock pan shares many of the benefits of cast iron but is lighter, cheaper, easier to clean, and the pan heats up quickly too, but at a controlled setting, for excellent browning and flavourful crust development on food.